Deformation and Bonding in a Puckered ReⴚC Square Net
Erika F. Merschrod S.
a)
, Aliya Courtney
b)
and Roald Hoffmann*
Ithaca, New York/U.S.A., Cornell University, Department of Chemistry and Chemical Biology and Cornell Center for Materials
Research
Received April 23
rd
, 2002.
Dedicated to Professor Rudolf Hoppe on the Occasion of his 80th Birthday
Abstract. Sc
5
Re
2
C
7
contains unusual Re⫺C puckered “square”
nets, isolated C
3
units, and scandium ions.Its electron count drives
the peculiar distortion of the Re⫺C nets, resulting in quite different
Re⫺CandRe⫺Re bonding along each direction. Indeed, some of
the Re⫺C interactions emerge as stronger than double bonds.The
puckering, in turn, enables a bonding role for the scandium atoms
Deformation und Bindung in einem gefalteten quadratischen ReⴚC-Netz
Inhaltsübersicht. Sc
5
Re
2
C
7
enthält ungewöhnliche „quadratische“
Re⫺C-Netze, isolierte C
3
-Einheiten und Scandium-Ionen. Die An-
zahl seiner Elektronen bestimmt die besondere Verzerrung der
Re⫺C-Netze, was ganz verschiedene Re⫺C- und Re⫺Re-Bindun-
gen in jeder Richtung zur Folge hat. Tatsächlich erscheinen einige
Introduction
Ternary transition metal carbides exhibit great structural
diversity, containing isolated carbon atoms, C
2
pairs and
C
3
groups. One also finds in them interesting metal-carbon
networks of varying dimensionality. A decade ago, a new
carbide was synthesized by Pöttgen and Jeitschko [1] which
contains both C
3
groups and individual C atoms. Sc
5
Re
2
C
7
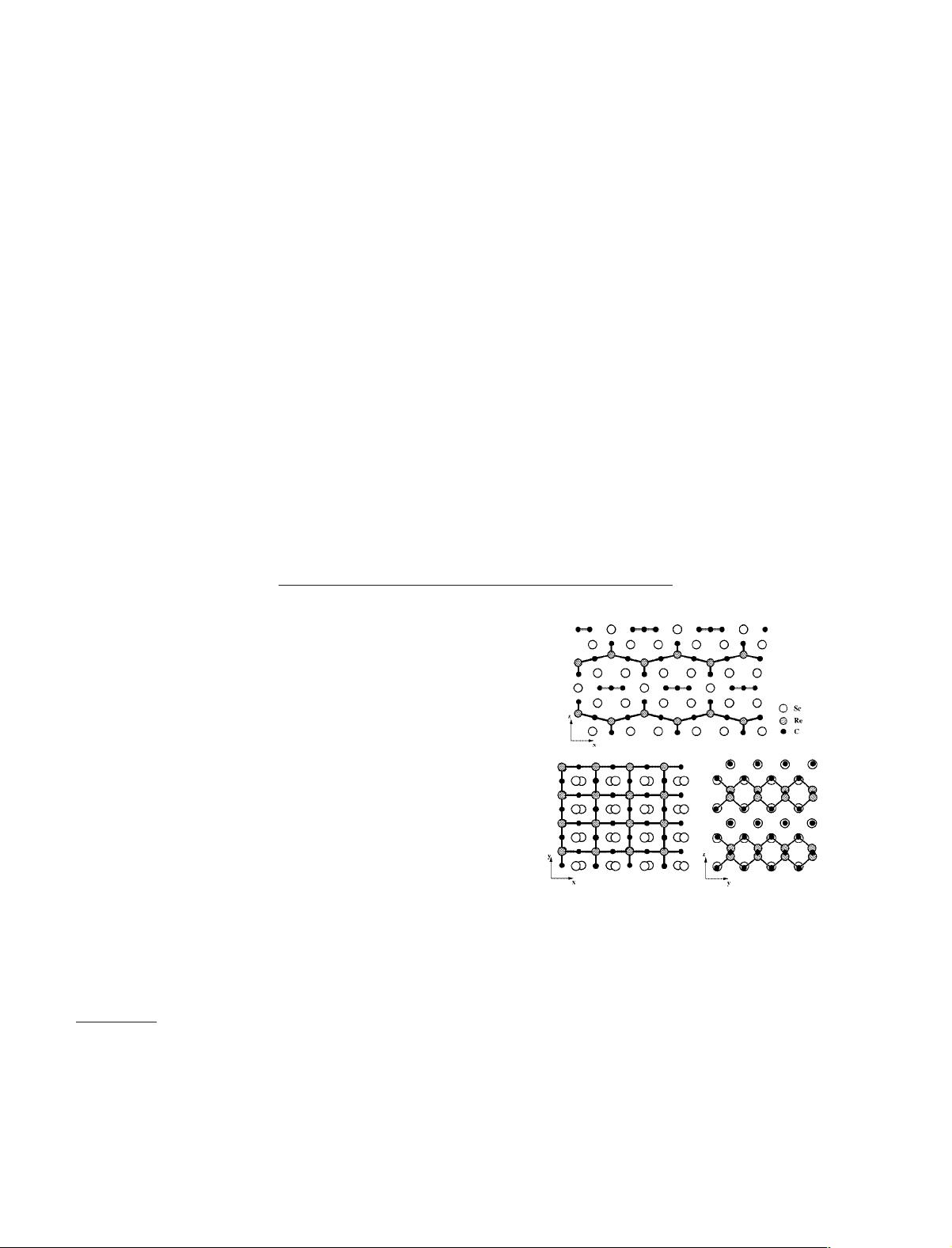
(shown in three views in 1) has square nets of rhenium and
carbon atoms which are highly distorted through pucker-
ing; the bonds in these nets are indicated with heavier black
lines. The axes in 1 are assigned for convenience during
analysis (placing the nets in the xy plane); our z axis corre-
sponds to the b axis of the original compound.
The Re⫺C sheets have two types of carbon atoms (small
black circle), each bridging two Re atoms (lined circles) in
either a linear or a bent geometry. The linear C
3
units are
isolated from each other and from the Re⫺C nets by the
scandium atoms (large white circles), which also separate
* Prof. Roald Hoffmann
Baker Laboratory, Department of Chemistry and Chemical
Biology, Cornell University
Ithaca, NY 14853-1301/U.S.A.
a)
Present address: Department of Chemistry, University of
Toronto, Toronto, Ontario, M5S 3H6, Canada
b)
Present address: Johns Hopkins University School of Medicine,
Baltimore, Maryland, 21205-2196, U.S.A.
Z. Anorg. Allg. Chem. 2002, 628, 2757⫺2763 2002 WILEY-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim 0044⫺2313/02/628/2757⫺2763 $ 20.00⫹.50/0 2757
in addition to their function as electron donors to the nets and the
isolated carbide units.
Keywords: Electronic structure; Extended Hückel theory; Tran-
sition-metal carbide: Scandium bonding; Metal⫺carbon multiple
bond
der Re⫺C-Wechselwirkungen stärker als Doppelbindungen. Die
Faltung erlaubt den Scandium-Atomen andererseits ihre Rolle als
Bindungspartner zusätzlich zu ihrer Funktion als Elektronen-Do-
noren des Netzes und der isolierten Carbid-Einheiten.
1
the two-dimensional Re⫺C networks from each other. The
unusual Re⫺C square nets attracted our interest, leading to
the investigation of the electronic structure of the com-
pound reported in this paper.
Assigning electrons
We begin by dissecting the three-dimensional structure of
Sc
5
Re
2
C
7
into its chemically meaningful components and
distributing the electrons accordingly. Based on the results









