image noise. This removed the need for the application of an inho-
mogeneity correction algorithm prior to data processing and
thus simplified the analysis of the results, since no consideration
of the inhomogeneity correction was needed in the analysis of
the performance of the algorithm. However, several authors
(e.g. MacDonald et al., 2000) have presented cortical thickness
measurements performed on inhomogeneity corrected data. This
implies that the algorithm presented here will be applicable to
other MR image types following the application of a suitable cor-
rection algorithm. The scanning protocols used are typical of scans
acquired clinically, in that the in-plane resolution is sub-millime-
tre, whereas the through-plane resolution is of the order of a few
millimetres, in order to increase the signal to noise ratio of the
data. From a purely geometrical perspective, if it is possible to
show that the method presented below produces reliable results
when applied to such data, then there is no reason to expect that
it will not work on more research-oriented protocols that may
have isotropic voxels: the reverse may not be the case, hence the
use here of the clinical scans. Reducing the slice thickness will
inevitably reduce the signal-to-noise ratio, but this will manifest
itself as an increase in the random errors on the results, rather than
the introduction of a systematic error.
2.2. Data pre-processing
The first of the data pre-processing stages involved application
of the partial volume segmentation algorithm described by Pokric
et al. (2001). The algorithm fitted a model consisting of Gaussian
distributions for each pure tissue and uniform distributions, con-
volved with Gaussian distributions representing the noise process,
for each partial volume class, to the intensity histogram of the im-
age volume. This resulted in an estimate of the mean and standard
deviation of each pure tissue for use in later stages of the cortical
thickness measurement algorithm. The histogram was constructed
using voxel intensities within a block of the brain containing only
WM, GM and CSF, manually defined in the Talairach coordinates
and then transformed into the coordinate system of each image
volume using the result of an affine registration. The position of
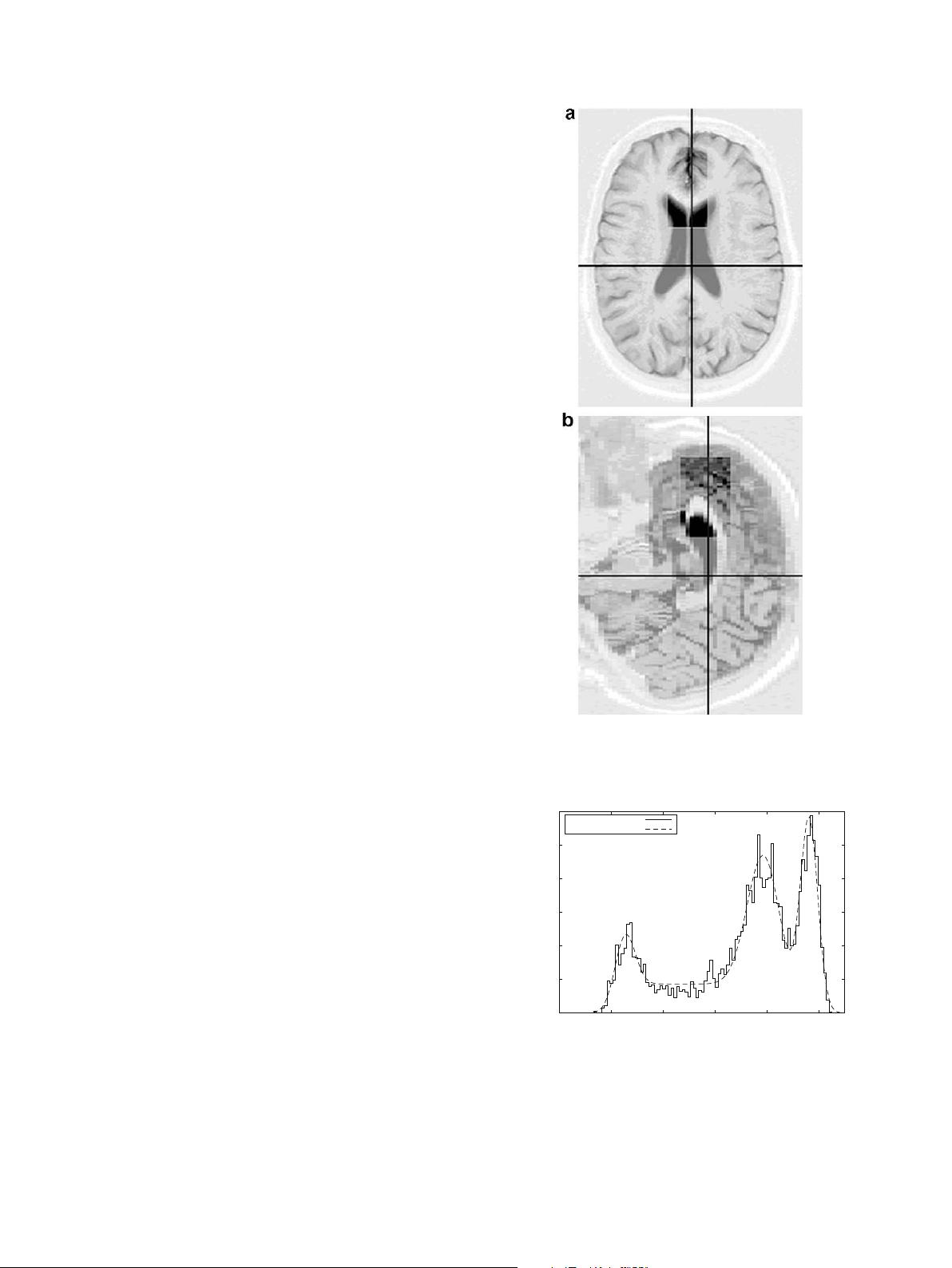
the block is illustrated in Fig. 2. The positioning of the volume in
the frontal lobe was selected in order to obtain a sufficient number
of voxels of the three pure tissue types. In elderly brains, the differ-
entiation between all three tissue types becomes less distinct for
several reasons. The boundary between GM and WM appears much
more diffuse than in younger brains (the gradient between the two
becomes much more shallow) presumably due to demyelination of
the WM axons such that the WM appears more like the non-mye-
linated GM. In addition, the presence of fluid-filled lesions in WM
may result in the apparent presence of GM in the WM, and the
greater partial voluming of GM and CSF (due to increased CSF) re-
sults in the actual GM appearing to have anomalous (lower)
grey level values. In order to obtain realistic estimates of the pure
tissue values, CSF values were taken from the ventricles, WM from
the corpus callosum, where there did not appear to be much
CSF/WM partial voluming, and GM from the cortex in front of the
anterior cingulate, with relatively little CSF/GM partial voluming.
This resulted in a histogram with well-defined peaks (see Fig. 3),
which could be fitted in order to calibrate the mean tissue values.
Although not strictly necessary for algorithmic purposes, it was
desirable to increase the degree of isotropy of the voxel dimensions
in order to allow a single step size to be used in the search along
the normal to the inner cortical surface, as described below,
regardless of position within the image volume. Therefore, the data
were up interpolated in the through-plane direction by a factor of
two. The partial volume grey level analysis described above al-
lowed estimation of the fractional volumetric contributions of
pairs of tissues to partial volume voxels. This identifies a set of pos-
sible boundary locations within the voxel, differing in which
surfaces of the voxel they intersect with. The most probable
boundary location was then selected from amongst these possibil-
ities according to the best agreement with the adjacent slices.
Fig. 2. Sections illustrating the approximate position of the cuboid, here shown in
greater contrast than the surrounding tissue, from which the three tissue histogram
is constructed: (a) axial view, (b) sagittal view.
0
10
20
30
40
50
60
-2500 -2000 -1500 -1000 -500 0
Number
Image Intensity Values
Data
Mixture Model
Fig. 3. Histogram of voxel intensity values (solid line) and fit (dashed line) of the
Gaussian mixture model to the histogram. There are clear peaks for the cerebro-
spinal fluid, grey matter and white matter at approximately 1800, 500 and 100
grey levels, respectively.
272 M.L.J. Scott et al. / Medical Image Analysis 13 (2009) 269–285









