Delayed Fluorescence in a Solution-Processable Pure Red Molecular
Organic Emitter Based on Dithienylbenzothiadiazole: A Joint Optical,
Electroluminescence, and Magnetoelectroluminescence Study
Ping Chen,
†,§
Li-Ping Wang,
‡
Wan-Yi Tan,
‡
Qi-Ming Peng,
†
Shi-Tong Zhang,
†
Xu-Hui Zhu,*
,‡
and Feng Li*
,†
†
State Key Laboratory of Supramolecular Structure and Materials, Jilin University, Changchun, 130012, China
‡
Institute of Polymer Optoelectronic Materials and Devices, State Key Laboratory of Luminescent Materials and Devices, South
China University of Technology, Guangzhou 510640, China
§
School of Physical Science and Technology, MOE Key Laboratory on Luminescence and Real-Time Analysis, Southwest University,
Chongqing 400715, P. R. China
*
S
Supporting Information
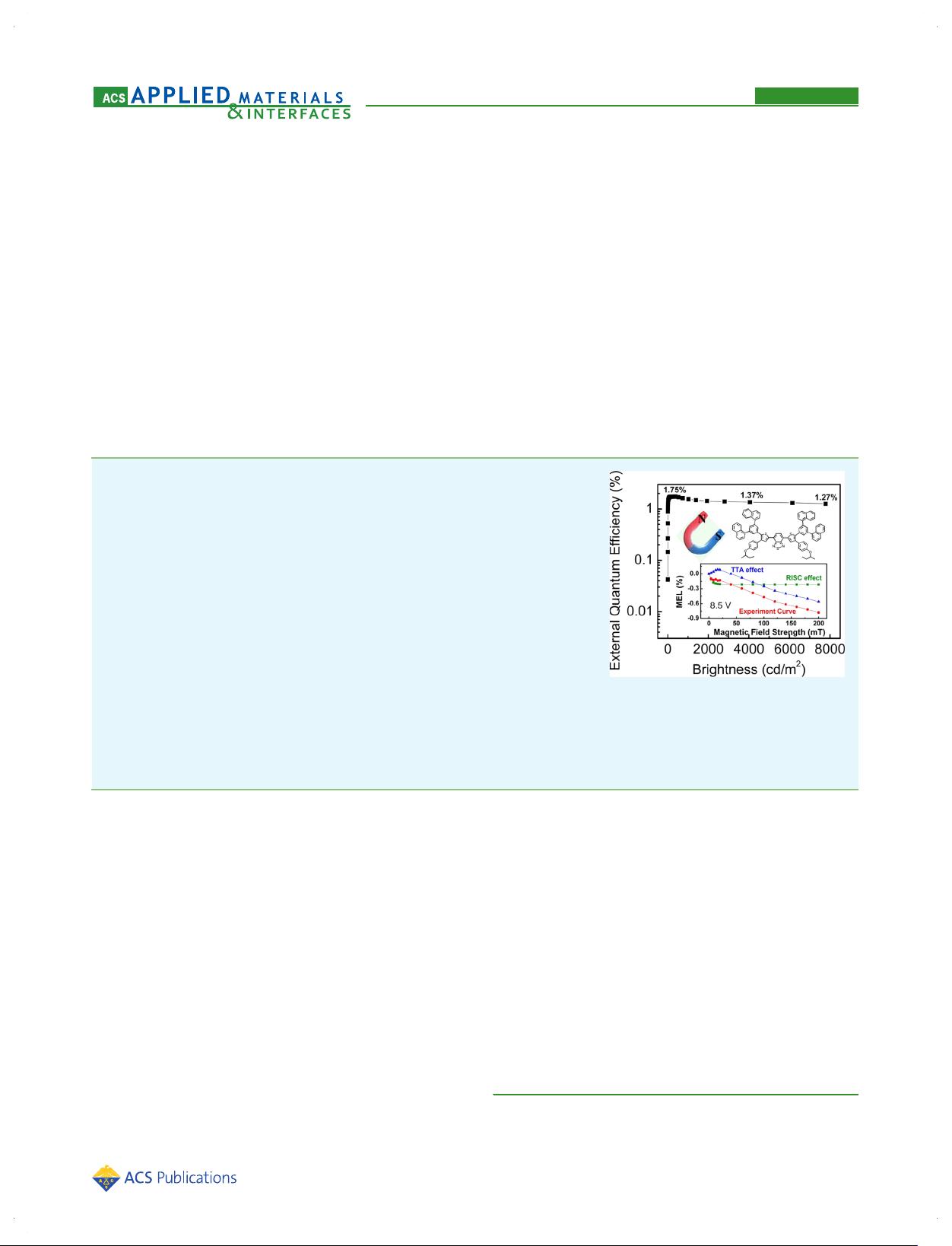
ABSTRACT: The discovery of triplet excitons participating in the photoluminescent
processes in a growing number of pure organic emitters represents an exciting
impetus for a diversity of promising opto, bio, and optoelectronic applications. In this
contribution, we have studied a small-molecule dithienylbenzothiadiazole-based red-
emitting dye red-1b, which shows clearly delayed fluorescence under optical and
electrical excitation. The OLED device that contained red-1b as a nondoped solution-
processable emitter exhibited a moderately high utilization of exciton amounting to
≈31% and slow efficiency roll-off. Magnetoelectroluminescence measurements
revealed the coexistence of reverse intersystem crossing from the lowest triplet
state to singlet state (RISC, E-type triplet to singlet up-conversion) and triplet−triplet
annihilation (TTA, P-type triplet to singlet up-conversion). Specifically, in low
current-density regime, the moderately high exciton utilization is attributed to RISC
(i.e., thermally activated delayed fluorescence, TADF), whereas in high current-
density regime, TTA may contribute to suppressing efficiency roll-off. Furthermore,
the results showed that red-1b may represent a new kind of organic red emitters that display delayed fluorescence in a way
differing from the few red emitters investigated so far.
KEYWORDS: exciton utilization, reverse intersystem crossing, triplet−triplet annihilation, electroluminescence,
magnetoelectroluminescence
1. INTRODUCTION
Organic fluorophores exhibiting delayed fluorescence have
gained very recently a growing momentum in organic light-
emitting diodes (OLEDs),
1−5
electrogenerated chemilumines-
cence,
6
and time-resolved fluorescence imaging.
7
Consequently,
they may eventually provide a low-cost alternative to
phosphorescent complexes that contain expensive and scarce
metals, in particular for high-performance OLED displays and
solid-state lighting.
Delayed fluorescence can stem from three different
processes: (i) reverse intersystem crossing (RISC) from the
lowest triplet to singlet excited state (E-type T → S up-
conversion, or thermally activated delay fluorescence);
8
(ii)
triplet−trip let anni hilation (T TA, P-t ype T → Sup-
conversion),
9
and (iii) geminate electron−hole recombina-
tion.
10
Nevertheless, in RISC, it has been shown by theoretical
calculation that a higher triplet excited state may potentially be
involved, leading to the so-called “hot exciton” mechanism,
provided that the energy barrier between T
1
→ S
1
is sufficiently
high.
4,5
Owing to the enormous prospects, a considerable number of
organic emitters displaying E-type and/or P-type delayed
fluorescence have been reported, revealin g rich chemical
structure−property relationships.
1,11−15
Remarkably, Adachi et
al. disclosed high-efficiency OLEDs by utilizing thermally
activated delay fluorescence materials (TADF, E-type) that
were previously achievable only by phosphorescent metal
complexes, amounting to a near unity of internal quantum
efficiency.
16
In li ght of these advances, we repor t herein delayed
fluorescence in a pure red molecular emitter based on
dithienylbenzothiadiazole, denoted as red-1b.
17
A combined
optical, electroluminescence (EL), and magnetoelectrolumines-
Received: December 8, 2014
Accepted: January 13, 2015
Published: January 13, 2015
Research Article
www.acsami.org
© 2015 American Chemical Society 2972 DOI: 10.1021/am508574m
ACS Appl. Mater. Interfaces 2015, 7, 2972−2978









