17
03 May 2019
Ling Wang
(852) 2800 8599
ling.wang@jpmorgan.com
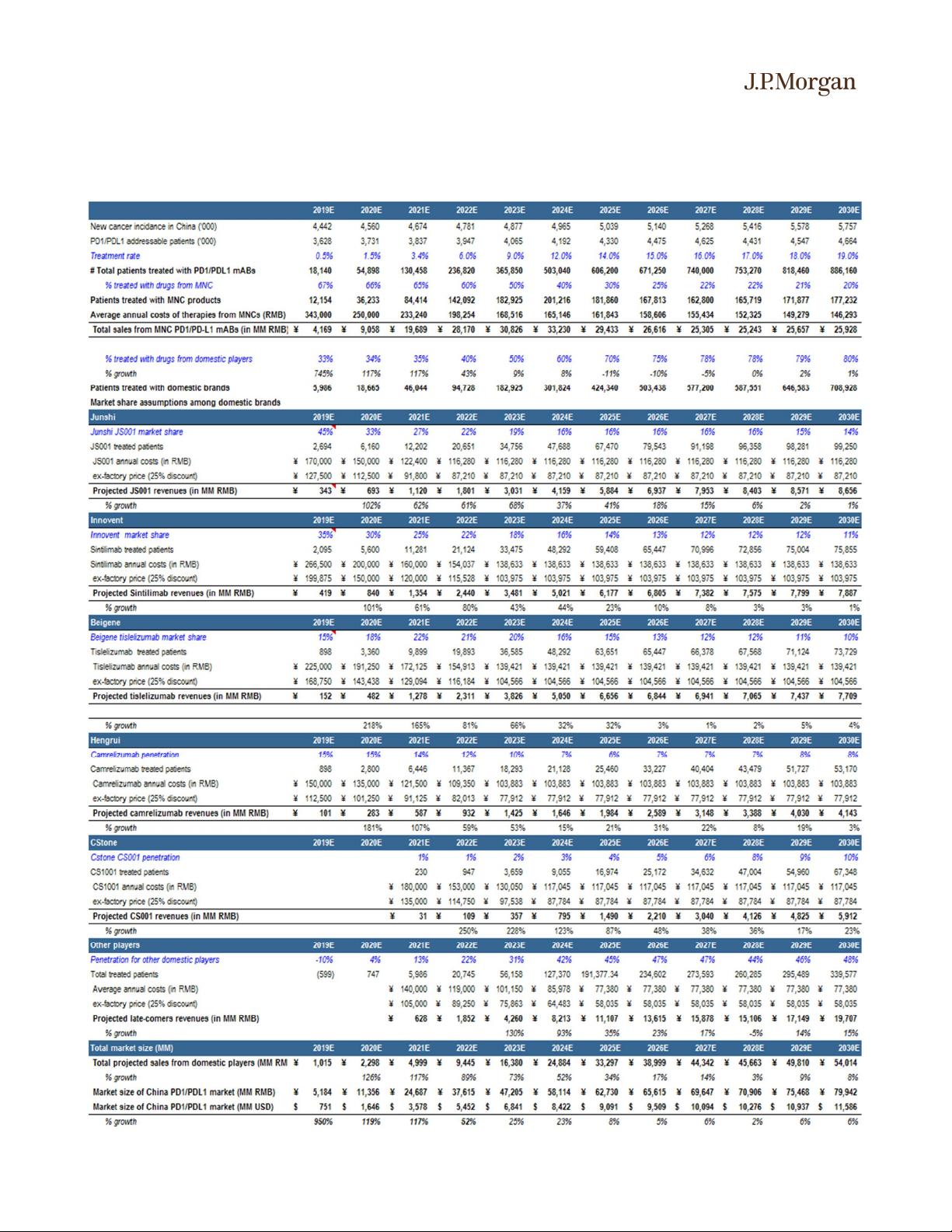
However, over the long run, with the increased body of clinical data from
domestic PD-1/PD-L1 mABs and pricing advantage, we expect the domestic
players to take significant market share while MNCs’ products are likely to be
reserved for high-end patients. Based on our model, by 2030, domestic players
are expected to occupy 2/3 of the total sales whereas MNCs are expected to only
have 1/3% of the market share.
The market share assumptions among domestic players will largely depend on a
number of factors: 1) evolving clinical profile (efficacy and safety) of the
molecule in large indications; 2) pricing strategy of the products; 3) the timing for
the drug to get approved / reimbursed in large indications; 4) the sale &
marketing capability of the team.
Investment risks
Generally speaking, the future of a high growth sector like China biotechnology,
which has many changing opportunities is by nature associated with many
uncertainties. In the near to intermediate term, we believe execution risks, clinical,
regulatory and marketing risks are the key risks of investing in biopharmaceutical
companies in China. Since the emergence of the China biopharmaceutical industry is
heavily driven by the China government’s favorable policy support, accompanied by
huge opportunity are significant risks. In our view, over the longer term, the main
risks in investing in the China biopharmaceutical industry are the uncertainties
surrounding the policies and macroeconomic factors. Additionally, the US/China
trade war may also impact the sustainable growth of the industry since collaborations
between Chinese companies and innovative biotech companies and top academic
centers in the US are likely an important source of future innovations. More
specifically, investing in the companies are also subject to the following risks:
Clinical risk and regulatory risk
Drug development and clinical trials carry inherent risk. Potential negative data or
unexpected delays in these trials and clinical programs from other trials, as well as
potential delays in obtaining regulatory approval will generate significant downside
pressure for the shares. Among the four companies we cover, Junshi and Innovent’s
PD-1 mABs have obtained regulatory approval in their initial indications while
BeiGene and CStone’s lead compounds have not been approved. Additionally, all of
these PD-1/PD-L1 mABs are still being evaluated in multiple late stage clinical trials
in many expanded indications with much larger market opportunity than their initial
indications.
Commercial risk
The market opportunities of the PD-1/PD-L1 products and the companies’ other
products may not be as large as we expected. In China, since the government
reimbursement is likely the largest payor of the novel therapeutics, the pricing of
drugs is expected to drop order to be included in NRDL. So far, despite price being a
potential headwind in China, we see the discount pricing model as rational, given
that volume growth appears to be more than offsetting the price cuts.
For the PD-1/PD-L1 market, there will be competition with MNCs as well as
domestic players. In particular, if MNCs decide to drop the prices of their PD-1/PD-
L1 substantially, it would negatively impact the market opportunities of domestic
products. Additionally, Henrui's PD-1 mAB is under review by CDE. Currently there