没有合适的资源?快使用搜索试试~ 我知道了~
首页fundamental of physics
fundamental of physics
fundamental physics textbook for engineering college sutdents
资源详情
资源评论
资源推荐

chapter
Electric Fields
P UZZLER
Soft contact lenses are comfortable to
wear because they attract the proteins in
the wearer’s tears, incorporating the
complex molecules right into the lenses.
They become, in a sense, part of the
wearer. Some types of makeup exploit
this same attractive force to adhere to
the skin. What is the nature of this force?
(Charles D. Winters)
Chapter Outline
23.1 Properties of Electric Charges
23.2 Insulators and Conductors
23.3 Coulomb’s Law
23.4 The Electric Field
23.5 Electric Field of a Continuous
Charge Distribution
23.6 Electric Field Lines
23.7 Motion of Charged Particles in a
Uniform Electric Field
P UZZLER
708

23.1 Properties of Electric Charges 709
he electromagnetic force between charged particles is one of the fundamen-
tal forces of nature. We begin this chapter by describing some of the basic
properties of electric forces. We then discuss Coulomb’s law, which is the fun-
damental law governing the force between any two charged particles. Next, we in-
troduce the concept of an electric field associated with a charge distribution and
describe its effect on other charged particles. We then show how to use
Coulomb’s law to calculate the electric field for a given charge distribution. We
conclude the chapter with a discussion of the motion of a charged particle in a
uniform electric field.
PROPERTIES OF ELECTRIC CHARGES
A number of simple experiments demonstrate the existence of electric forces and
charges. For example, after running a comb through your hair on a dry day, you
will find that the comb attracts bits of paper. The attractive force is often strong
enough to suspend the paper. The same effect occurs when materials such as glass
or rubber are rubbed with silk or fur.
Another simple experiment is to rub an inflated balloon with wool. The bal-
loon then adheres to a wall, often for hours. When materials behave in this way,
they are said to be electrified, or to have become electrically charged. You can eas-
ily electrify your body by vigorously rubbing your shoes on a wool rug. The electric
charge on your body can be felt and removed by lightly touching (and startling) a
friend. Under the right conditions, you will see a spark when you touch, and both
of you will feel a slight tingle. (Experiments such as these work best on a dry day
because an excessive amount of moisture in the air can cause any charge you build
up to “leak” from your body to the Earth.)
In a series of simple experiments, it is found that there are two kinds of elec-
tric charges, which were given the names positive and negative by Benjamin
Franklin (1706–1790). To verify that this is true, consider a hard rubber rod that
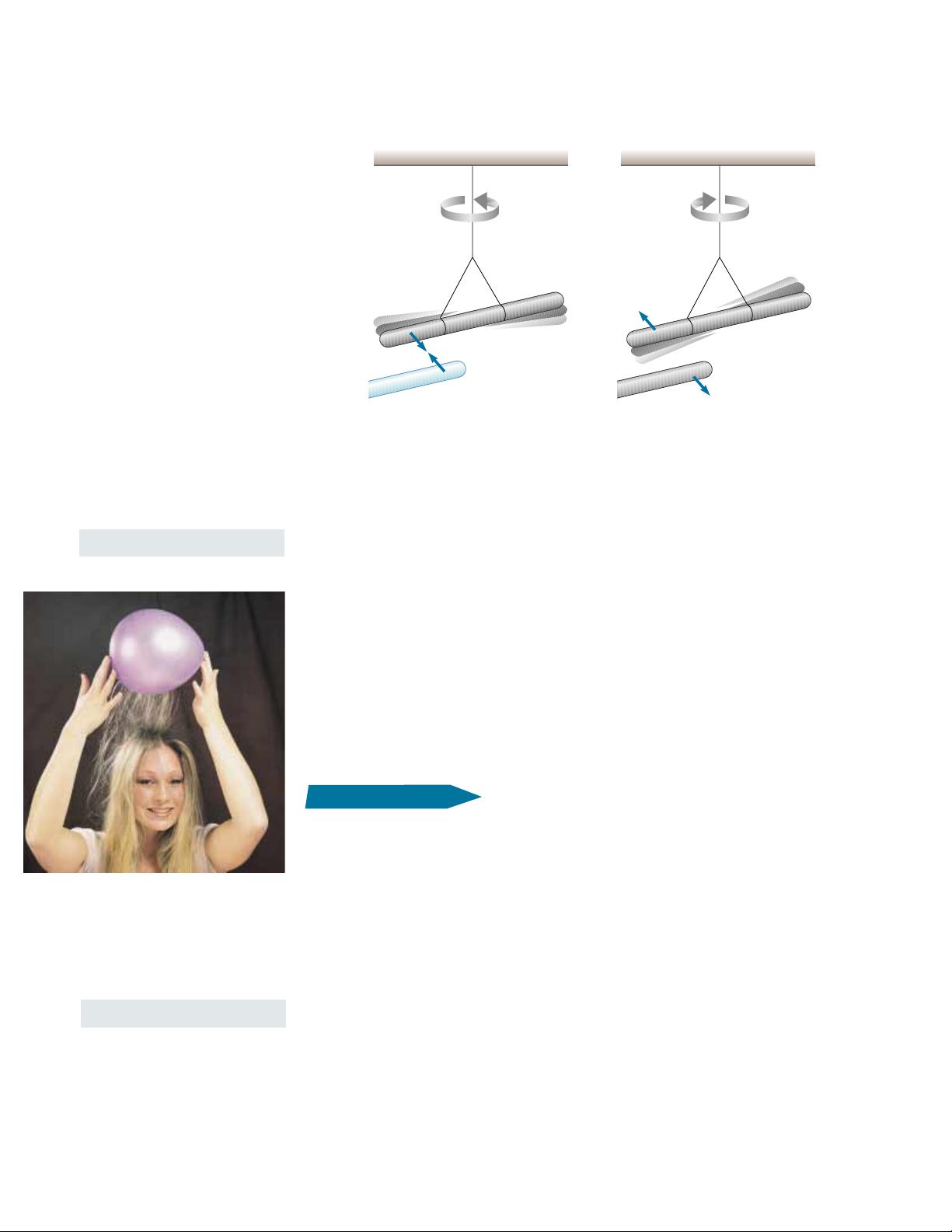
has been rubbed with fur and then suspended by a nonmetallic thread, as shown
in Figure 23.1. When a glass rod that has been rubbed with silk is brought near the
rubber rod, the two attract each other (Fig. 23.1a). On the other hand, if two
charged rubber rods (or two charged glass rods) are brought near each other, as
shown in Figure 23.1b, the two repel each other. This observation shows that the
rubber and glass are in two different states of electrification. On the basis of these
observations, we conclude that like charges repel one another and unlike
charges attract one another.
Using the convention suggested by Franklin, the electric charge on the glass
rod is called positive and that on the rubber rod is called negative. Therefore, any
charged object attracted to a charged rubber rod (or repelled by a charged glass
rod) must have a positive charge, and any charged object repelled by a charged
rubber rod (or attracted to a charged glass rod) must have a negative charge.
Attractive electric forces are responsible for the behavior of a wide variety of
commercial products. For example, the plastic in many contact lenses, etafilcon, is
made up of molecules that electrically attract the protein molecules in human
tears. These protein molecules are absorbed and held by the plastic so that the
lens ends up being primarily composed of the wearer’s tears. Because of this, the
wearer’s eye does not treat the lens as a foreign object, and it can be worn com-
fortably. Many cosmetics also take advantage of electric forces by incorporating
materials that are electrically attracted to skin or hair, causing the pigments or
other chemicals to stay put once they are applied.
23.1
T
11.2
QuickLab
Rub an inflated balloon against your
hair and then hold the balloon near a
thin stream of water running from a
faucet. What happens? (A rubbed
plastic pen or comb will also work.)
710 CHAPTER 23 Electric Fields
Another important aspect of Franklin’s model of electricity is the implication
that electric charge is always conserved. That is, when one object is rubbed
against another, charge is not created in the process. The electrified state is due to
a transfer of charge from one object to the other. One object gains some amount of
negative charge while the other gains an equal amount of positive charge. For ex-
ample, when a glass rod is rubbed with silk, the silk obtains a negative charge that
is equal in magnitude to the positive charge on the glass rod. We now know from
our understanding of atomic structure that negatively charged electrons are trans-
ferred from the glass to the silk in the rubbing process. Similarly, when rubber is
rubbed with fur, electrons are transferred from the fur to the rubber, giving the
rubber a net negative charge and the fur a net positive charge. This process is con-
sistent with the fact that neutral, uncharged matter contains as many positive
charges (protons within atomic nuclei) as negative charges (electrons).
If you rub an inflated balloon against your hair, the two materials attract each other, as
shown in Figure 23.2. Is the amount of charge present in the balloon and your hair after
rubbing (a) less than, (b) the same as, or (c) more than the amount of charge present be-
fore rubbing?
In 1909, Robert Millikan (1868–1953) discovered that electric charge always
occurs as some integral multiple of a fundamental amount of charge e. In modern
terms, the electric charge q is said to be quantized, where q is the standard symbol
used for charge. That is, electric charge exists as discrete “packets,” and we can
write where N is some integer. Other experiments in the same period
showed that the electron has a charge e and the proton has a charge of equal
magnitude but opposite sign e. Some particles, such as the neutron, have no
charge. A neutral atom must contain as many protons as electrons.
Because charge is a conserved quantity, the net charge in a closed region re-
mains the same. If charged particles are created in some process, they are always
created in pairs whose members have equal-magnitude charges of opposite sign.
q Ne,
Quick Quiz 23.1
Rubber
Rubber
(a)
F
F
(b)
F
F
Rubber
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
+
+
+
+
+
+
Glass
–
+
Figure 23.1 (a) A negatively charged rubber rod suspended by a thread is attracted to a posi-
tively charged glass rod. (b) A negatively charged rubber rod is repelled by another negatively
charged rubber rod.
Figure 23.2 Rubbing a balloon
against your hair on a dry day
causes the balloon and your hair
to become charged.
Charge is conserved
Charge is quantized

23.2 Insulators and Conductors 711
From our discussion thus far, we conclude that electric charge has the follow-
ing important properties:
• Two kinds of charges occur in nature, with the property that unlike charges
attract one another and like charges repel one another.
• Charge is conserved.
• Charge is quantized.
Properties of electric charge
INSULATORS AND CONDUCTORS
It is convenient to classify substances in terms of their ability to conduct electric
charge:
23.2
Electrical conductors are materials in which electric charges move freely,
whereas electrical insulators are materials in which electric charges cannot
move freely.
Materials such as glass, rubber, and wood fall into the category of electrical insula-
tors. When such materials are charged by rubbing, only the area rubbed becomes
charged, and the charge is unable to move to other regions of the material.
In contrast, materials such as copper, aluminum, and silver are good electrical
conductors. When such materials are charged in some small region, the charge
readily distributes itself over the entire surface of the material. If you hold a cop-
per rod in your hand and rub it with wool or fur, it will not attract a small piece of
paper. This might suggest that a metal cannot be charged. However, if you attach a
wooden handle to the rod and then hold it by that handle as you rub the rod, the
rod will remain charged and attract the piece of paper. The explanation for this is
as follows: Without the insulating wood, the electric charges produced by rubbing
readily move from the copper through your body and into the Earth. The insulat-
ing wooden handle prevents the flow of charge into your hand.
Semiconductors are a third class of materials, and their electrical properties
are somewhere between those of insulators and those of conductors. Silicon and
germanium are well-known examples of semiconductors commonly used in the
fabrication of a variety of electronic devices, such as transistors and light-emitting
diodes. The electrical properties of semiconductors can be changed over many or-
ders of magnitude by the addition of controlled amounts of certain atoms to the
materials.
When a conductor is connected to the Earth by means of a conducting wire or
pipe, it is said to be grounded. The Earth can then be considered an infinite
“sink” to which electric charges can easily migrate. With this in mind, we can un-
derstand how to charge a conductor by a process known as induction.
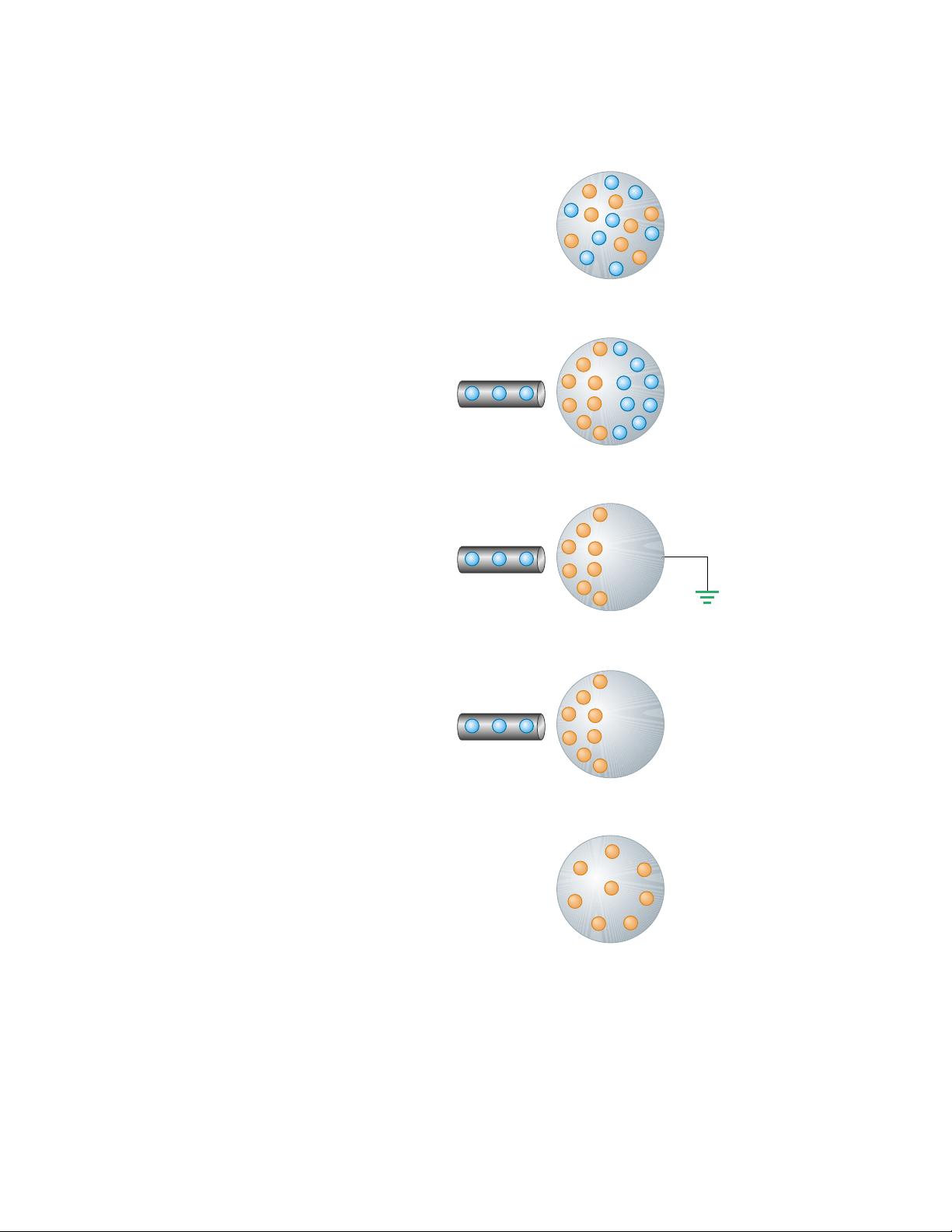
To understand induction, consider a neutral (uncharged) conducting sphere
insulated from ground, as shown in Figure 23.3a. When a negatively charged rub-
ber rod is brought near the sphere, the region of the sphere nearest the rod ob-
tains an excess of positive charge while the region farthest from the rod obtains an
equal excess of negative charge, as shown in Figure 23.3b. (That is, electrons in
the region nearest the rod migrate to the opposite side of the sphere. This occurs
even if the rod never actually touches the sphere.) If the same experiment is per-
formed with a conducting wire connected from the sphere to ground (Fig. 23.3c),
some of the electrons in the conductor are so strongly repelled by the presence of
11.3
Metals are good conductors
Charging by induction
712 CHAPTER 23 Electric Fields
+
+
+
+
+
+
+
+
–
–
–
–
–
–
–
–
–– –
(b)
+
+
+
+
+
+
+
+
–– –
(c)
+
+
+
+
+
+
+
+
–– –
(d)
+
+
+
+
+
+
(e)
+
+
+
+
+
+
+
+
–
–
–
–
–
–
–
–
(a)
+
+
Figure 23.3 Charging a metallic object by induction (that is, the two objects never touch each
other). (a) A neutral metallic sphere, with equal numbers of positive and negative charges.
(b) The charge on the neutral sphere is redistributed when a charged rubber rod is placed near
the sphere. (c) When the sphere is grounded, some of its electrons leave through the ground
wire. (d) When the ground connection is removed, the sphere has excess positive charge that is
nonuniformly distributed. (e) When the rod is removed, the excess positive charge becomes uni-
formly distributed over the surface of the sphere.
剩余403页未读,继续阅读
hz21157
- 粉丝: 0
- 资源: 1
上传资源 快速赚钱
 我的内容管理
收起
我的内容管理
收起
 我的资源
快来上传第一个资源
我的资源
快来上传第一个资源
 我的收益 登录查看自己的收益
我的收益 登录查看自己的收益 我的积分
登录查看自己的积分
我的积分
登录查看自己的积分
 我的C币
登录后查看C币余额
我的C币
登录后查看C币余额
 我的收藏
我的收藏  我的下载
我的下载  下载帮助
下载帮助

会员权益专享
最新资源
- c++校园超市商品信息管理系统课程设计说明书(含源代码) (2).pdf
- 建筑供配电系统相关课件.pptx
- 企业管理规章制度及管理模式.doc
- vb打开摄像头.doc
- 云计算-可信计算中认证协议改进方案.pdf
- [详细完整版]单片机编程4.ppt
- c语言常用算法.pdf
- c++经典程序代码大全.pdf
- 单片机数字时钟资料.doc
- 11项目管理前沿1.0.pptx
- 基于ssm的“魅力”繁峙宣传网站的设计与实现论文.doc
- 智慧交通综合解决方案.pptx
- 建筑防潮设计-PowerPointPresentati.pptx
- SPC统计过程控制程序.pptx
- SPC统计方法基础知识.pptx
- MW全能培训汽轮机调节保安系统PPT教学课件.pptx
资源上传下载、课程学习等过程中有任何疑问或建议,欢迎提出宝贵意见哦~我们会及时处理!
点击此处反馈



安全验证
文档复制为VIP权益,开通VIP直接复制
 信息提交成功
信息提交成功
评论2