mediate binding to the cell surface and hence determine
cell speci®city [15,16]. The envelope fuses with the cell
membrane releasing the virion contents into the cyto-
plasm. A discrete number of different host cell surface
proteins can serve as receptors for the retroviral envelope
proteins. Receptors, such as CD4 which binds lentivirus,
are expressed by cells of speci®c lineages such as
lymphocytes, while amphotropic, xenotropic and gibbon
ape leukemia virus (GALV) receptors which bind MMLV
are expressed on most, if not all, mammalian cells.
Retroviral virions that express different envelope pro-
teins, such as VSV-G, have increased stability as well as an
expanded host range [17]. This is primarily due to the
ability of the VSV-G to recognize membrane phospholipid
as a minimal receptor [18,19]. Unlike standard murine
retroviruses, VSV-G retrovirus vectors are also relatively
resistant to deactivation by human complement [20], can
be concentrated to high titers by centrifugation, and
frozen/thawed without loss of infectivity [17]. Viral
tropism has also been altered by generating a protease-
cleavable, receptor-blocking domain onto the viral coat
protein. Exposure to a speci®c protease that can cleave
the site is suf®cient to restore virus infectivity at sites with
high local activity of the relevant protease [21]. Other
means used to target retrovirus vectors to speci®c cell
types include: chemical modi®cation, use of antibody
ligands, and creation of recombinant envelope proteins
(for a review see [22]).
Receptor-mediated entry of adenovirus into cells is
dependent on two of its coat proteins, the ®ber and
penton base. The ®ber mediates primary attachment to a
wide range of cell types via the Coxsackie-adenovirus
receptor (CAR) protein [23]. Following attachment, an
Arg-Gly-Asp (RGD) tripeptide motif in the penton base
protein binds to a
v
integrins on the host cell membrane,
which then trigger cellular internalization by receptor-
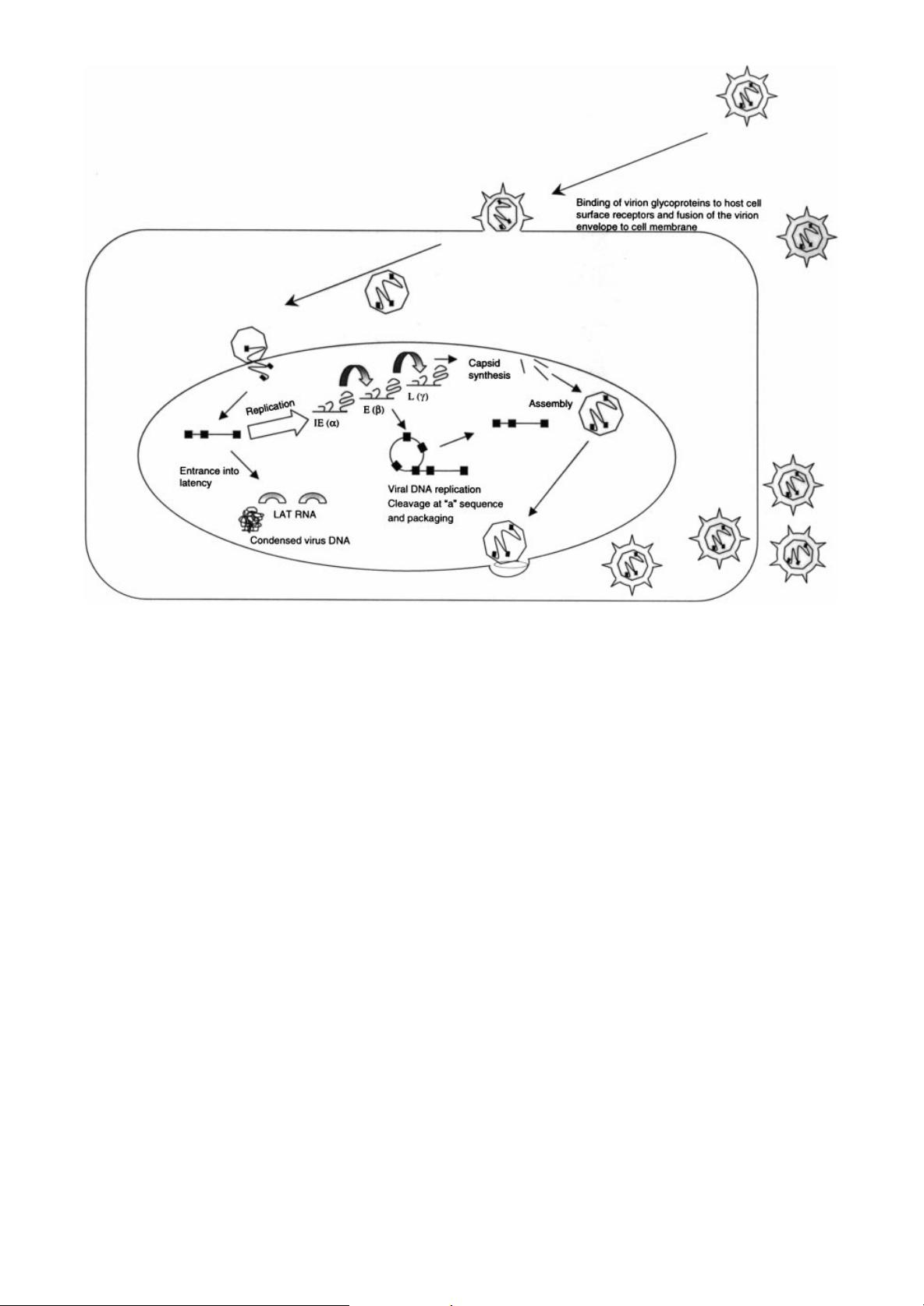
Figure 1. Schematic view of HSV infection. Glycoproteins in the envelope of the virion mediate binding to the cell surface and
fusion with the plasma membrane. The capsid containing the viral DNA is transported to the nuclear membrane via an active
microtubular mechanism. The capsid opens and extrudes the virus DNA into the nucleus through nuclear pores. Within the
nucleus, wild-type HSV can either replicate to produce more virus particles or enter latency. Virus replication involves a cas-
cade of virus gene expression involving sequential transcription of immediate early (IE), early (E) and late (L) genes. Viral
DNA is replicated as a continuum and enters newly formed, empty capsids via binding of the `a' or pac sequence. When the
capsid is full the virus DNA is cut at the pac sequence and the capsid seals closed. Capsids acquire an envelope via transport
through the nuclear and plasma membranes (for a review see [140]). Ultimately production of HSV virions leads to cell death.
Under some conditions, e.g. low MOI in sensory neurons, the virus DNA enters an episomal state of latency in the cell nucleus
characterized by a tight con®guration and production of latency associated transcripts (LATs) which is benign, but from which
the virus can reactivate to productive replication
Hybrid Viral Vector Designs 397
Copyright # 2000 John Wiley & Sons, Ltd. J Gene Med 2000; 2: 395±408.









