Duan et al. ASD With Machine Learning
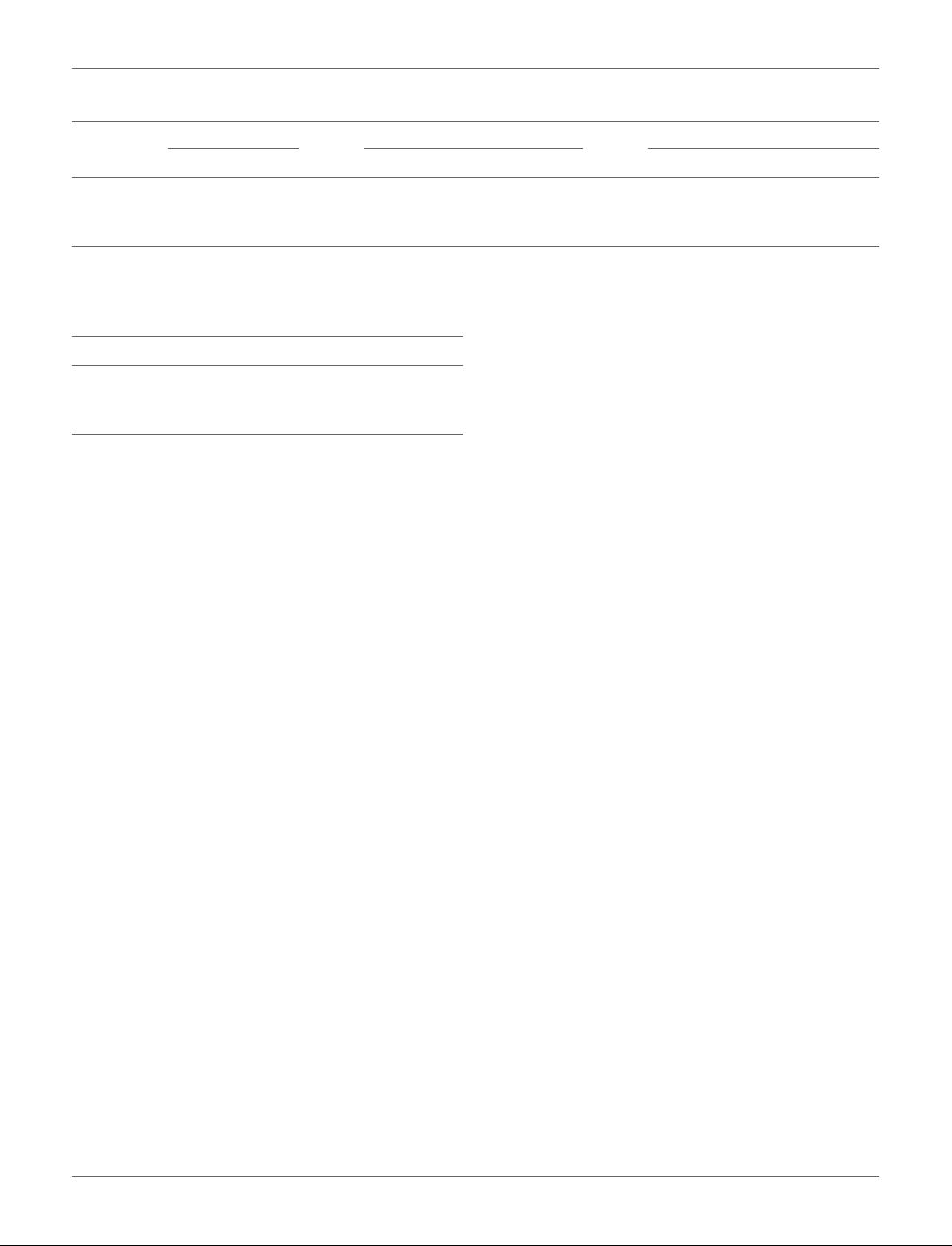
TABLE 1 | Demographics information.
Site Gender(M/F) Age Full scale IQ
NC ASD NC ASD NC ASD
GU 28/27 43/8 10.448 ± 1.696 10.896 ± 1.535 121.46 ± 13.808 118.3 ± 15.377
OHSU 27/29 30/7 10.38 ± 1.636 11.81 ± 2.271 117.46 ± 11.968 105.97 ± 16.734
UCLA 11/5 15/1 9.3 ± 2.09 11.13 ± 2.247 115 ± 13.05 102.06 ± 13.959
TABLE 2 | The scanning parameters of structural MRI imaging in Georgetown
University (GU), Oregon Health and Science University (OHSU), and University of
California Los Angeles (UCLA) with Siemens.
Site Voxel size(mm
3
) Flip angle(deg) FOV(mm) TR(ms) TE(ms) T1(ms)
GU 1× 1 × 1 7 256 × 256 2530 3.5 1100
OHSU 1× 1 v 1.1 10 256 × 256 2300 3.58 900
UCLA 1× 1 × 1.2 9 256 × 256 2300 2.86 853
http://www.filion.ucl.ac.uk/spm/software/spm8/) and the VBM8
(Voxel-Based Morphometry) toolbox (http://dbm.neuro.uni-
jena.de/vbm) running under Matlab R2014a (Mathworks). At
first, all T1-weighted images were corrected for bias-field
inhomogeneities and then segmented into GM, WM, and CSF
(cerebrospinal fluid) based on a tissue probability map (
Mazziotta
et al., 1 995). The segmented GM/WM image was spatially
normalized to the “IXI500_MNI152” template based on t h e
DARTEL algorithm (Ashburner, 2007). After that, non-linear
warping for t he effect of spatial normalization was corrected
to generate GM/WM modulated normalized images. Finally,
spatial smoothing (Gaussian k ernel with 6 mm full-width at half-
maximum) was conducted on GM/WM images to remove noise.
2.3. Statistical Analysis
In the present study, a two-sample t-test has been employed
on the GU dataset with age, gender, Total Intracranial Volume
(TIV) as t h e effect-of-no-interest covariates to identify group
differences between ASD and TDC. A significance level of p <
0.001 (uncorrected) was established with an extent threshold of
50 voxels. Meanwhile, an absolute threshold mask of 0.1 was used
on GM/WM volume images to avoid potential edge effe c ts.
3. METHODS
This study aimed to identify the brain abnormality and predict
ASD from TDC via machine learning techniques. However,
neuroimaging-based ML models like the “black-box” and unable
to be understood from the prospect of neuroscience. To
address this issue,
Kohoutov et al. (2020) has developed a
unified framework to enhance the interpretability of ML models
and provide mechanistic insights into underlying neural or
disease processes. The proposed framework contains a three-
stage process of assessment including Model-level assessment,
Feature-level assessment, and Biology-level assessment. In the
first stage, the ML model has been built from observations and
assessed in terms of its sensitivity, specificity, and generalizability.
In the second stage, significant features have been identified
from a prediction within the model. Finally, the neuroscientific
plausibility of the ML model has been proved with evidence from
previous literature and other studies.
However, ML models based on neuroimaging are often built
on numerous features and limited participants, which makes t he
model is prone to overfitting and leads to poor generalization
and expensive comput ational cost even if dimensional reduction
techniques have been used. Moreover, isolated features are
often insufficient to acquire satisfactory predictive performance
and explain the model performance. Consequently, th e study
has designed a neighborhood-to-regional machine learning
workflow within this unified framework to identify structural
alteration and discriminant ASD from TDC. The workflow
proposed in the study has been illustrated in Figure 1.
3.1. Model-Level Assessment
First, the study has built an ML model based on the searchlight
technique (
Kriegeskorte et al., 2006). A spherical window is
centered at each voxel to generate a data matrix from the
voxel and its neighbors. In light of the spherical window,
PCA(Principal Component Analysis) has been used to reduce
the dimensionality of the matrix, and SVM(Support Vector
Machine) has been used to achieve the classification.
3.1.1. Principal Component Analysis
Supposed data matrix obtained from training data x =
{x
1
, x
2
, ...x
m
} ∈ R
m×n
is obtained from a spherical window,
where m is the number of subjects in the training dataset, n
represents the voxel number centered a specific voxel within
a spherical window, PCA (
Wold et al., 1987) has been used
to reduce the dimensionality of the matrix by transforming
high-dimensional data into lower-dimensional features while
preser ving its maximum variance. To this end, data points are
projected from high-dimensional space to low-dimensional space
with the following linear combinations:
y =
n
X
j=1
a
j
x
j
= Xa (1)
where a = {a
1
, a
2
, ..., a
n
} ∈ R
n×k
and k ≪ n, y is the
low-dimensional features. Meanwhile, the variance of the low-
dimensional fe at ure is given by:
var(Xa) = a
T
Sa (2)
Frontiers in Human Neuroscience | www.frontiersin.org 4 February 2022 | Volume 15 | Article 765517

 我的内容管理
收起
我的内容管理
收起
 我的资源
快来上传第一个资源
我的资源
快来上传第一个资源
 我的收益 登录查看自己的收益
我的收益 登录查看自己的收益 我的积分
登录查看自己的积分
我的积分
登录查看自己的积分
 我的C币
登录后查看C币余额
我的C币
登录后查看C币余额
 我的收藏
我的收藏  我的下载
我的下载  下载帮助
下载帮助 


 信息提交成功
信息提交成功
评论0