
contribution
1
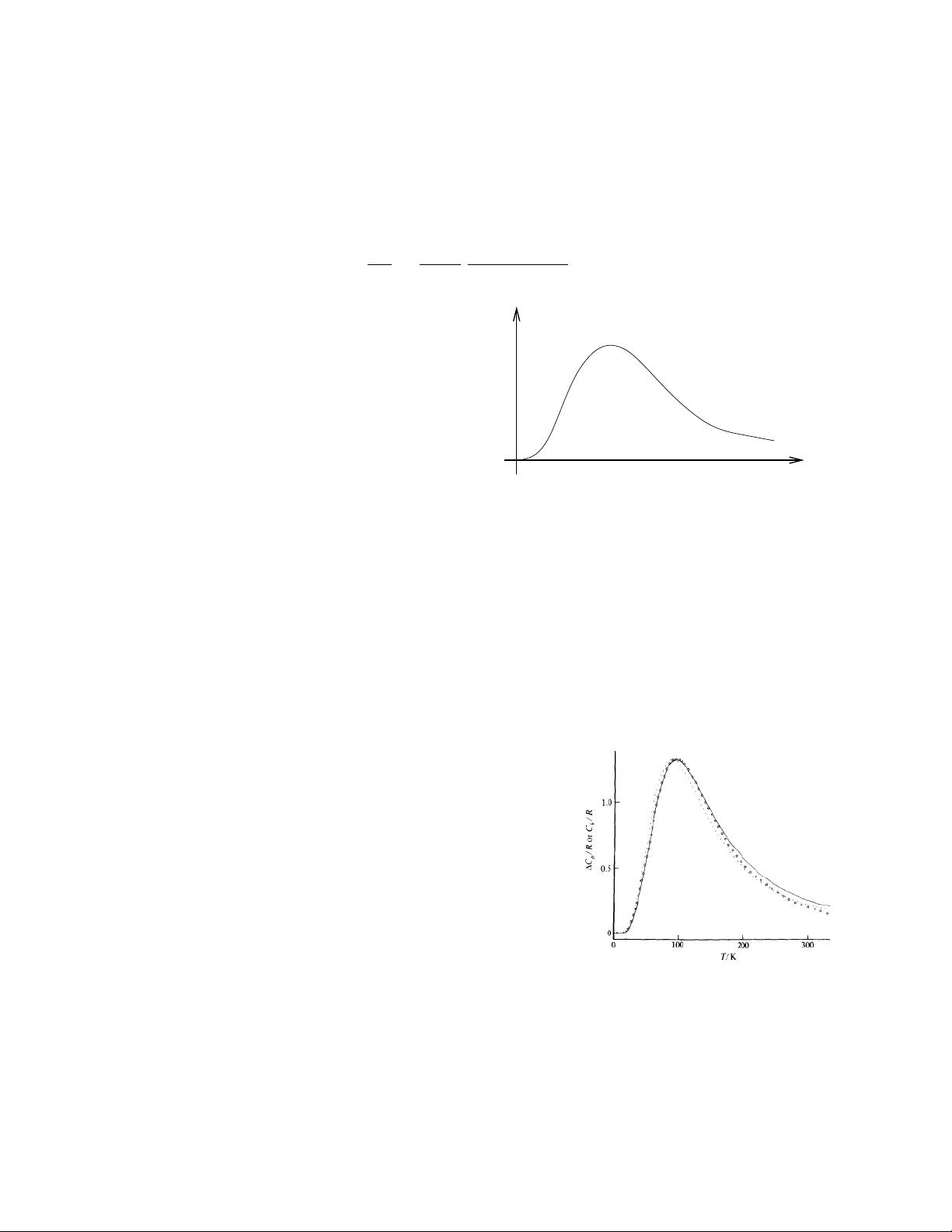
. The open circles and dots are both data (interpreted in mildly different
ways); the solid line is theoretical prediction, albeit complicated slightly by the presence
of a number of spin states. The deviations are most likely due to interactions between
the spins.
The two state system can also be used as a model for defects in a lattice. In this case,
the “spin down” state corresponds to an atom sitting in the lattice where it should be
with no energy cost. The “spin up” state now corresponds to a missing atom, ejected
from its position at energy cost .
1.2.4 Pressure, Volume and the First Law of Thermodynamics
We will now start to consider other external parameters which can affect different
systems. We will see a few of these as we move through the course, but the most
important one is perhaps the most obvious – the volume V of a system. This didn’t
play a role in the two-state example because the particles were fixed. But as soon
as objects are free to move about, it becomes crucial to understand how far they can
roam.
We’ll still use the same notation for the number of states and entropy of the system,
but now these quantities will be functions of both the energy and the volume,
S(E, V ) = k
B
log Ω(E, V )
The temperature is again given by 1/T = ∂S/∂E, where the partial derivative implicitly
means that we keep V fixed when we differentiate. But now there is a new, natural
quantity that we can consider — the differentiation with respect to V . This also gives
a quantity that you’re all familiar with — pressure, p. Well, almost. The definition is
p = T
∂S
∂V
(1.15)
To see that this is a sensible definition, we can replay the arguments of Section 1.2.2.
Imagine two systems in contact through a moveable partition as shown in the figure
above, so that the total volume remains fixed, but system 1 can expand at the expense
of system 2 shrinking. The same equilibrium arguments that previously lead to (1.8)
now tell us that the volumes of the systems don’t change as long as ∂S/∂V is the same
for both systems. Or, in other words, as long as the pressures are equal.
Despite appearances, the definition of pressure actually has little to do with entropy.
Roughly speaking, the S in the derivative cancels the factor of S sitting in T . To make
1
The data is taken from Chirico and Westrum Jr., J. Chem. Thermodynamics 12 (1980), 311, and
shows the spin contribution to the heat capacity of Tb(OH)
3
– 14 –


 信息提交成功
信息提交成功









