co-workers reported the same annulation reaction of
benzoic acids with alkynes using an electron-deficient
Rh(III) complex under ambient conditions (at RT–40
o
C), using AgNTf
2
and Cu(OAc)
2
·H
2
O as the
additives.
[9]
In 2013, Ison and co-workers developed a
relatively mild reaction conditions (60 °C, 24 h in
CH
3
OH), for the same annulation reaction using
(Cp*IrCl
2
)
2
catalyst and AgOAc as the oxidant.
[10]
Very recently, Sundararaju and co-workers reported a
nontoxic and air stable Cp*Co(CO)I
2
-catalyzed
annulation reaction of benzoic and acrylic acids with
alkynes for the synthesis of isocoumarins and pyrones.
In this reaction CuO was used as an oxidant and
2,2,2-trifluoroethanol (TFE) was used as a solvent.
[11]
In 2012, Ackermann and co-workers demonstrated
how the in-situ generated cationic Ru(II) catalyst
could be used for the annulation reaction of alkynes
with anilines, 5-aryl-1H-pyrazoles and oximes for the
efficient synthesis of indoles, pyrazolo[5,1-
a]isoquinoline and isoquinolines respectively
(Scheme 5).
[12]
Scheme 5. Ru(II)-Catalyzed synthesis of indoles,
pyrazolo[5,1-a]isoquinoline and isoquinolines
Li and co-workers developed an unprecedented
Ru(II)-catalyzed intramolecular annulation reaction of
amides with alkynes which proceeds via formyl
translocation for the synthesis of 1H-indole-3-
carbaldehydes (Scheme 6).
[13]
The reaction is assumed
to proceed via oxidative addition of Ru(II) in to the
N–CHO bond, followed by intramolecular
nucleophilic cyclization and reductive elimination
Scheme 6. Ru(II)-Catalyzed synthesis of 1H-indole-3-
carbaldehydes
pathways.
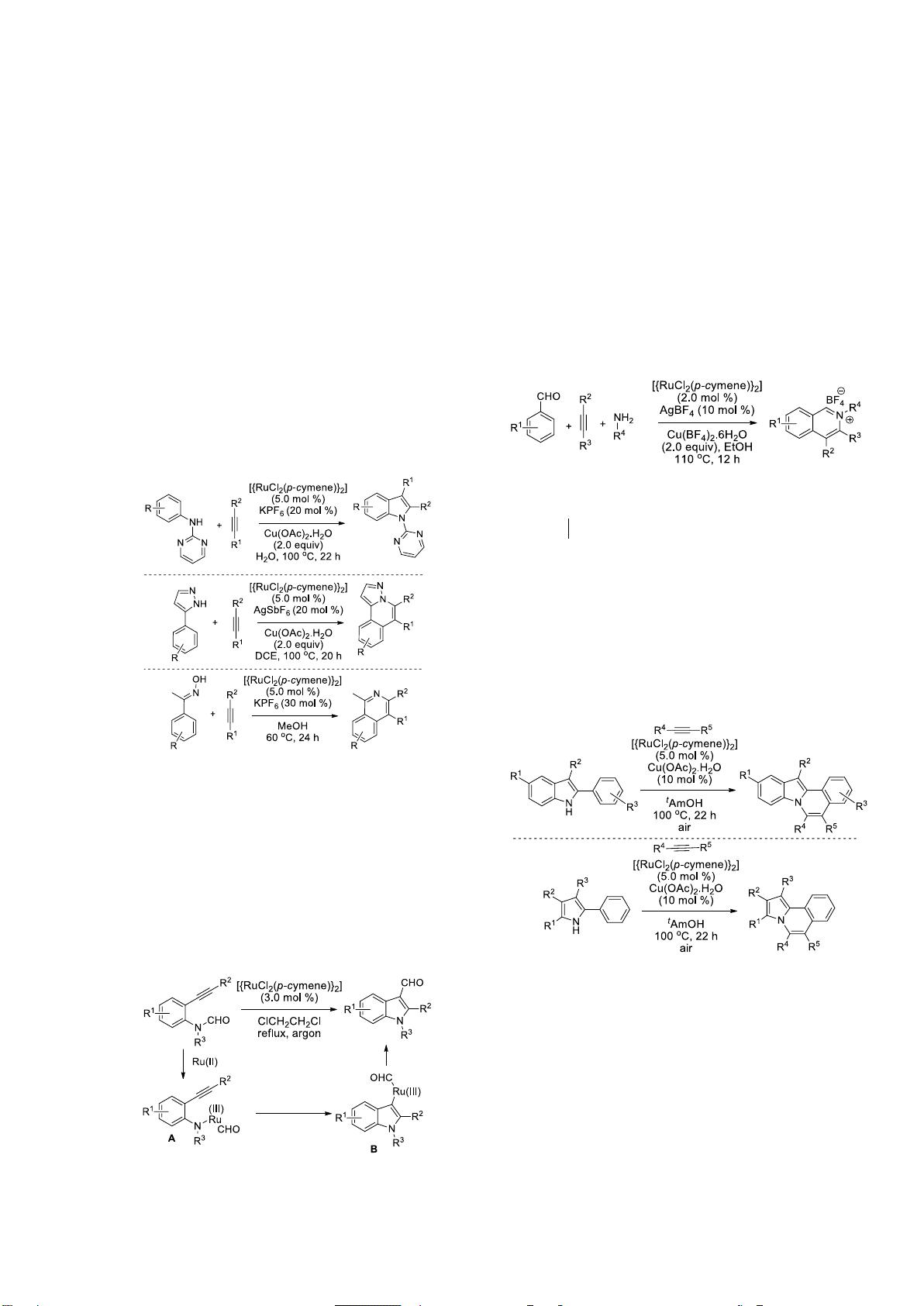
Cheng and co-workers developed a Ru(II)-
catalyzed C-H activation and annulation reaction of
benzaldehydes, amines and alkynes for the
regioselective synthesis of isoquinolinium salts
(Scheme 7).
[14]
A similar three-component reaction for
the synthesis of isoquinolinium salts was already
reported using expensive catalyst [{RhCp*Cl
2
}
2
] (2.0
mol %).
[15]
In the Ru-catalyzed reaction, the cationic
Ru complex first coordinates with the nitrogen atom
of the in situ generated imine. Then ortho-C-H
activation, alkyne insertion and finally reductive
elimination of the metal affords the isoquinolinium
salts. The metal is reoxidized to the active Ru(II) by
Cu(BF
4
)
2
.
Scheme 7. Ru(II)-
Catalyzed synthesis of isoquinolinium salts
Ackermann and co-workers developed a Ru(II)-
catalyzed C-H activation and annulation reaction of 2-
arylindole s with alkynes in the presence of catalytic
amount of Cu(OAc)
2
and air as the oxidants for the
synthesis of indolo[2,1-a]isoquinolines. They
extended the scope of the reaction for the annulation
of 2-arylpyrroles with alkynes which afforded good
yield of pyrrolo[2,1-a]isoquinolines (Scheme 8).
[16]
A
similar oxidative coupling reaction of 2-phenylindoles
with alkynes was reported by Miura and co-workers
for the synthesis of indolo[2,1-a]isoquinolines using
expensive [(Cp*RhCl
2
)
2
].
[17]
Scheme 8. Ru(II)-
Catalyzed synthesis of indolo[2,1-a]isoquinolines and
pyrrolo[2,1-a]isoquinolines.
Ackermann et al. described a Ru(II)-catalyzed
oxidative annulation reaction of electron rich
enamines with alkynes for the synthesis of pyrroles
utilizing air as the ideal oxidant (Scheme 9).
[18a]
Guan
and co-workers developed a Pd(OAc)
2
-catalyzed (5
mol %) annulation reaction of aryl enamides with
alkynes to afford triaryl- substituted pyrroles. The
reaction was performed using Cu(OAc)
2
(1.0 equiv)
as the oxidant and xantphos (10 mol %) as the ligand.
The electron-rich enamides which are challenging
Advanced Synthesis & Catalysis
This article is protected by copyright. All rights reserved.

 我的内容管理
展开
我的内容管理
展开
 我的资源
快来上传第一个资源
我的资源
快来上传第一个资源
 我的收益 登录查看自己的收益
我的收益 登录查看自己的收益 我的积分
登录查看自己的积分
我的积分
登录查看自己的积分
 我的C币
登录后查看C币余额
我的C币
登录后查看C币余额
 我的收藏
我的收藏  我的下载
我的下载  下载帮助
下载帮助 


 信息提交成功
信息提交成功