18 January & February 2018 www.elektormagazine.com
Although the Sand Clock [1] is super
cool, it has a few drawbacks for everyday
application. Due to the use of a sandbox,
it can only operate in the horizontal posi-
tion, which makes correct lighting essen-
tial for good legibility. The noise when
the sandbox is being shaken smooth is
unavoidable, but it can be very irritat-
ing — so the Sand Clock is not the ideal
choice for a clock on your bedside table
or nightstand.
The Laser Time Writer shows the time by
writing it with a violet laser on a piece of
self-adhesive phosphorescent (“glow in
the dark”) film. The written digits disap-
pear after a short while, depending on
the lighting conditions, allowing the cycle
to start over again. The physical back-
ground of this phenomenon is described
in more detail in the Intermezzo 1 inset.
We could have also used a bright white
or blue LED instead of the laser, but the
distance between the light source and
the phosphorescent film is less critical
with the laser — and it looks a lot more
high-tech.
If you want to convert an existing Sand
Clock into a Laser Time Writer, an
upgrade kit is available in the Elektor
Store. Figure 1 gives an impression of
the clock before and after the upgrade.
A full Laser Time Writer kit will also be
available in the near future — we’ll keep
you informed.
Phosphorescence in practice
In the past, zinc sulphide doped with cop-
per, or sometimes with silver, was used
as a phosphorescent material. Nowadays
alkaline earth metal aluminates doped
with europium, dysprosium or other rare
earth elements are used for this purpose.
More recently, doped alkaline-earth sili-
cates have been used for other colours,
such as blue. Most of these materials
are chemically and biologically inert and
therefore non-toxic.
As far as we know, the self-adhesive
film used here for the laser clock is only
available with green phosphorescence,
but if you want to experiment with other
colours you can buy a variety of “glow
in the dark” powders and use them to
make your own phosphorescent paints.
Suitable recipes can be found online.
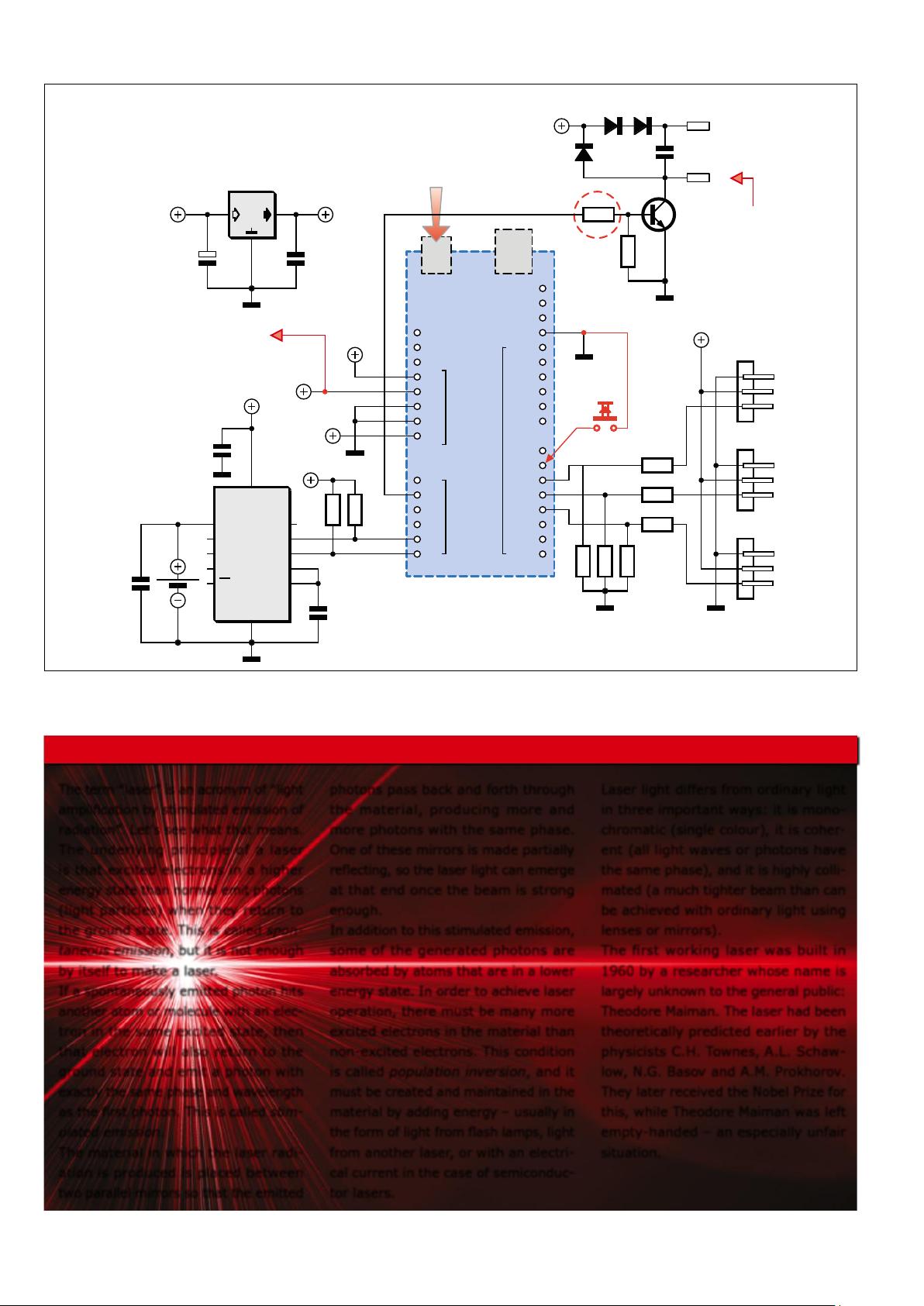
Mechanical construction
The mechanical construction of the
Laser Time Writer does not need much
description. It is largely based on the
Sand Clock, so conversion from the Sand
Intermezzo 1:
What’s glowing there in the dark?
The simple explanation is that “glow in
the dark” materials absorb energy from
light shining on them and then release
this energy over a relatively long period
in the form of light. This light emitted
over an extended time is usually green-
ish, but there are also materials that
emit other colours. As usual, the detailed
explanation is a bit more complicated.
Fluorescence and
phosphorescence
The general term for the emission of pho-
tons (light) by a material in response to
energy input into the material is lumines-
cence. However, the term glow is used
for materials that emit visible light when
strongly heated.
Photoluminescence is a form of lumines-
cence in which the energy is input in the
form of photons, causing electrons to
become excited. When these electrons
return to their ground state (which is
called decay), photons are emitted –
usually at a longer wavelength than
the incoming photons (this is called the
Stokes displacement).
Three different mechanisms can be dis-
tinguished for photoluminescence: flu-
orescence, phosphorescence, and the
Raman effect (which we will disregard
here).
With fluorescence, which is named after
the mineral fluorite (calcium fluoride) in
which this phenomenon was first stud-
ied, the time between the excitation and
decay of the electrons is very short (on
the order of nanoseconds), so imping-
ing light with a particular wavelength
results in the virtually immediate emis-
sion of light with another wavelength.
Some examples are dyes and pigments
that light up when exposed to ultraviolet
light (“black light”), fluorescent pens,
fluorescent vests, etc. Fluorescent tubes,
low-energy lamps, LED lamps and white
LEDs also use fluorescent substances —
which are confusingly called phosphors —
to convert ultraviolet or blue light into a
light spectrum that we perceive as white.
As early as the Middle Ages, materials
that emit light were called phosphors —
a name derived from the Greek word
phosphoros, which means “bearer of
light”. The chemical element phospho
-
rus, discovered by the German alchemist
Hennig Brand in 1669, owes its name
to the greenish glow emitted by white
phosphorus in contact with oxygen in the
air. However, this chemical reaction does
not have anything to do with phospho-
rescence. With phosphorescence, quan-
tum-mechanical effects delay the decay
of the electrons from the excited state
by times on the order of milliseconds to
hours, so phosphorescent materials con-
tinue to emit light after the initial light
source is removed. With glow-in-the-dark
materials this effect is especially strong,
and they can continue to “glow” (be vis-
ible in the dark) for hours after being
exposed to light.
Figure 1. The Sand Clock before the upgrade (left) and after the Laser Time Writer upgrade (right).