May 10, 2009 / Vol. 7, No. 5 / CHINESE OPTICS LETTERS 373
Experimental evaluation of rate coefficients for
Rb(5D
J
)+H
2
→ RbH+H reaction
Qian Wang (
)
1,2
, Yifan Shen (
)
2∗
, and Kang Dai (
ÜÜÜ
)
2
1
School of Science, Xi’an Jiaotong Universit y, Xi’an 710049, China
2
School of Physics Science and Technology, Xinjiang University, Urumqi 830046, China
∗
E-mail: shenyifan01@xju.edu.cn
Received June 12, 2008
The Rb(5D
J
)+H
2
→ RbH+H photochemical reaction has been studied. Rb vapor mixed with H
2
is
irradiated in a glass cell with 778-nm pulses which populate one of the 5
2
D states by two-photon absorption.
Measurements for the relative intensities of the atomic fluorescence and the absorption of the RbH product
near the axis of the cell yield the rate coefficients for the Rb(5D
3/2
)+H
2
and Rb(5D
5/2
)+H
2
reactions,
which are (3.6 ± 1.3) × 10
−11
and (1.7 ± 0.6) × 10
−11
cm
3
/s, respectively. The relative reactivity with H
2
for Rb(5D
3/2
) is higher than that for Rb(5D
5/2
).
OCIS codes: 020.0020, 020.2070.
doi: 10.3788/COL20090705.0373.
The collisions of excited alkali atoms with molecular hy-
drogen leading to a nonreactive inelastic scattering and
the reactive collision leading to the formation of metal hy-
dride have long been subjects of molecular dynamics
[1−9]
.
They represent the simplest three-body problems and
have been a very useful testing ground for a nonadia-
batic process and other dynamical models. Although the
collisions of excited alkali atoms with H
2
have been inves-
tigated for decades, there are still a limited number of sys-
tems for which one has both extended high-resolution ex-
perimental data and reliable theoretical calculations, and
the related reaction mechanism is not well understood
[10]
.
The reaction of Rb with H
2
has been far less studied than
the other alkali-hydrogen reactions since only a few ex-
perimental works have been published
[9,11]
.
In this letter, we present experimental results of the
reactive collisions between the Rb(5D
J
) state and H
2
molecules. It may be represented as
Rb(5D
J
)+H
2
→ RbH + H. (1)
Since the reaction (1) is exoergic by 0.4 eV, it is easy to
occur. We can determine the relative reactivity of the
Rb(5D
3/2
)andRb(5D
5/2
) atoms with H
2
by the mea-
surements for reaction rate coefficients.
When Rb atoms are selectively excited to one of the
5
2
D fine-structure states, its decay to the 5P
1/2
or 5P
3/2
state results in the emission of fluorescence. Before de-
caying, some of the primarily excited atoms may also
undergo reactive collisions causing the formation of the
ground state RbH molecules. Some others undergo in-
elastic collisions causing their transfer to the other 5
2
D
fine-structure state and radiationless transfer to other
lower-energy states. Therefore, in the Rb(5D
J
)-H
2
sys-
tem, in addition to the reactive process (1), there exist
the following energy transfer processes:
Rb(5D
3/2
)+H
2
←→ Rb(5D
5/2
)+H
2
, (2)
Rb(5D
J
)+H
2
→ Rb states other than Rb(5D)+H
2
. (3)
The processes (2) and (3) are nonreactive processes which
have influence on the reactive process (1).
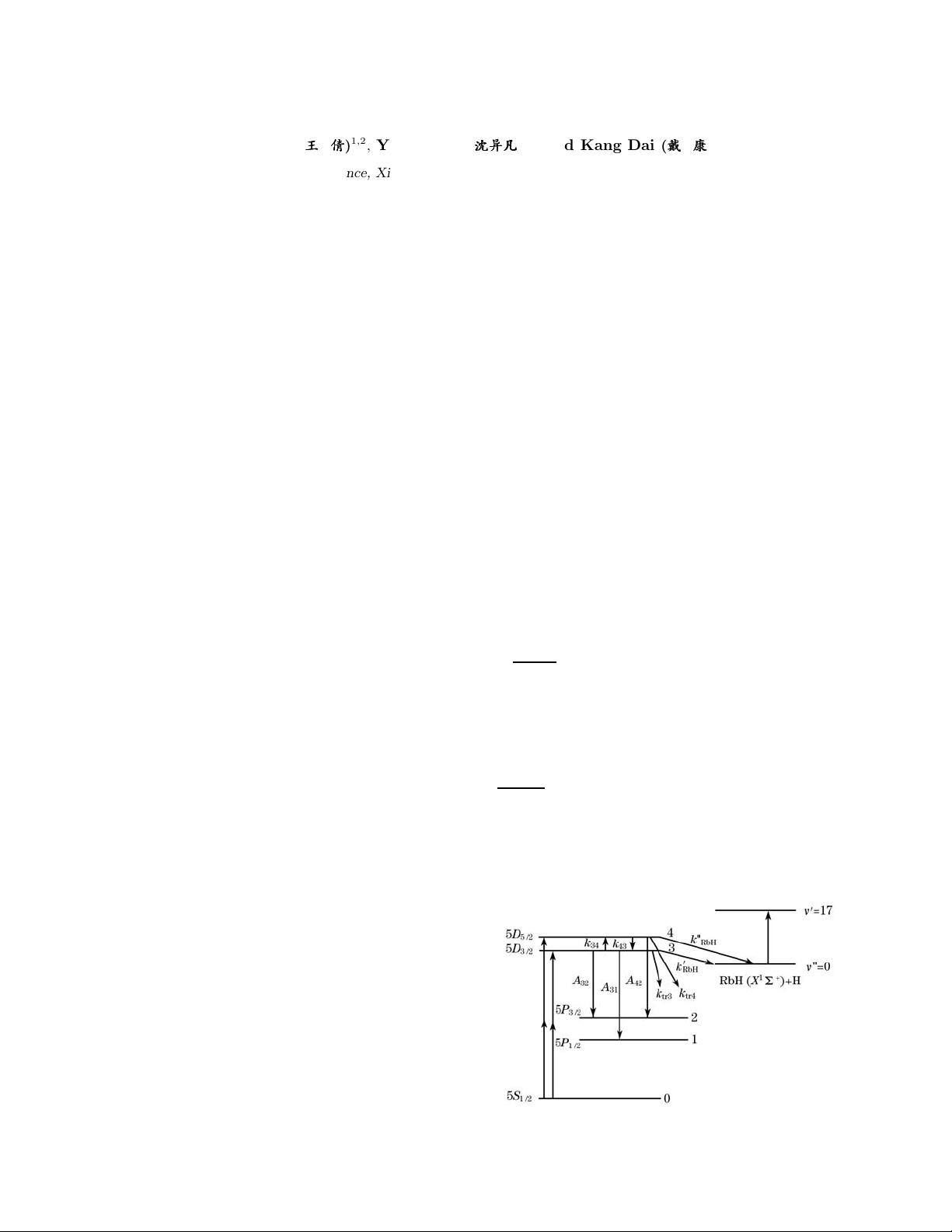
A partial energy-level diagram for rubidium, showing
the states involved in the experiment as well as the tran-
sitions between them, is shown in Fig. 1. In our exper-
iment, the Rb ground-state atom density in the vapor
phase was about 10
13
cm
−3[12]
while the density of H
2
mulecules was about 10
17
cm
−3
. The collisional energy
transfer due to the ground state rubidium may be ne-
glected.
When the 5D
3/2
state is optically excited, the popu-
lation density of the 5D
5/2
state may be represented by
the rate equation
dn
4
(t)
dt
= k
34
Nn
3
(t)
−(1/τ
4
+ k
4
N + k
43
N + k
tr4
N)n
4
(t).(4)
When the 5D
5/2
state is optically excited, the popu-
lation density of the 5D
3/2
state may be represented by
the rate equation
dn
3
(t)
dt
= k
43
Nn
4
(t)
− (1/τ
3
+ k
3
N + k
34
N + k
tr3
N) n
3
(t) .(5)
In Eqs. (4) and (5), τ
s
and n
s
(or n
s
) are lifetimes
and population densities of states with the subscripts
s =0− 4 corresponding to 5S
1/2
, 5P
1/2
, 5P
3/2
, 5D
3/2
,
Fig. 1. Energy diagram of the Rb(5D
J
)+H
2
reactive and non-
reactive energy transfer collisions.
1671-7694/2009/050373-04
c
2009 Chinese Optics Letters

 我的内容管理
展开
我的内容管理
展开
 我的资源
快来上传第一个资源
我的资源
快来上传第一个资源
 我的收益 登录查看自己的收益
我的收益 登录查看自己的收益 我的积分
登录查看自己的积分
我的积分
登录查看自己的积分
 我的C币
登录后查看C币余额
我的C币
登录后查看C币余额
 我的收藏
我的收藏  我的下载
我的下载  下载帮助
下载帮助 


 信息提交成功
信息提交成功